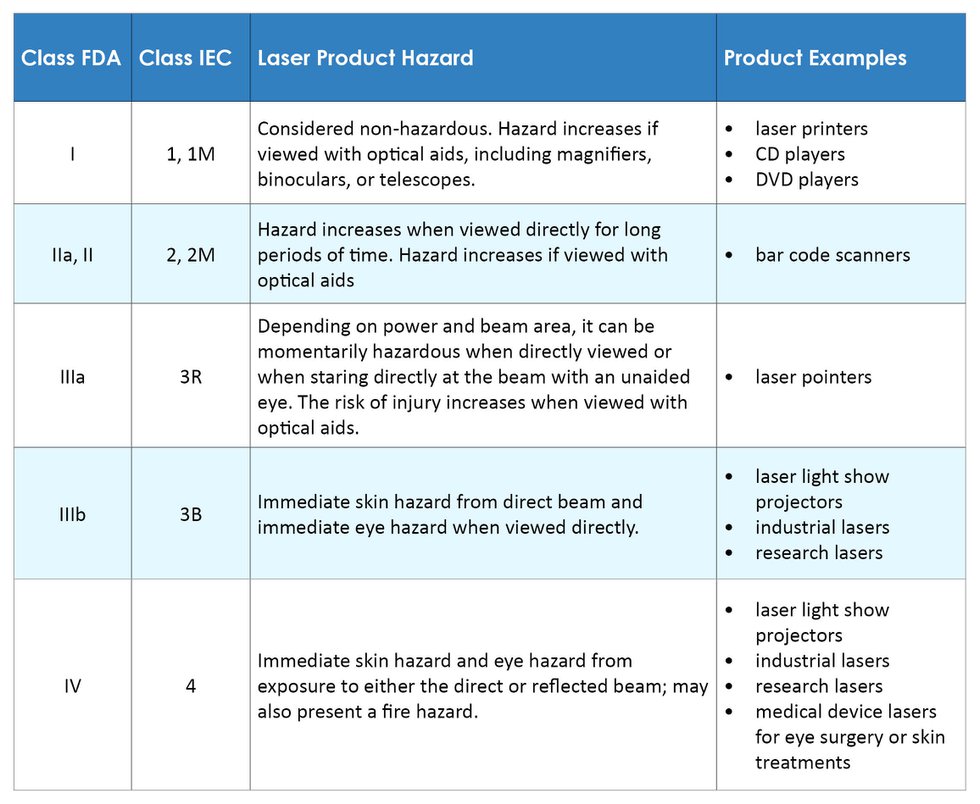
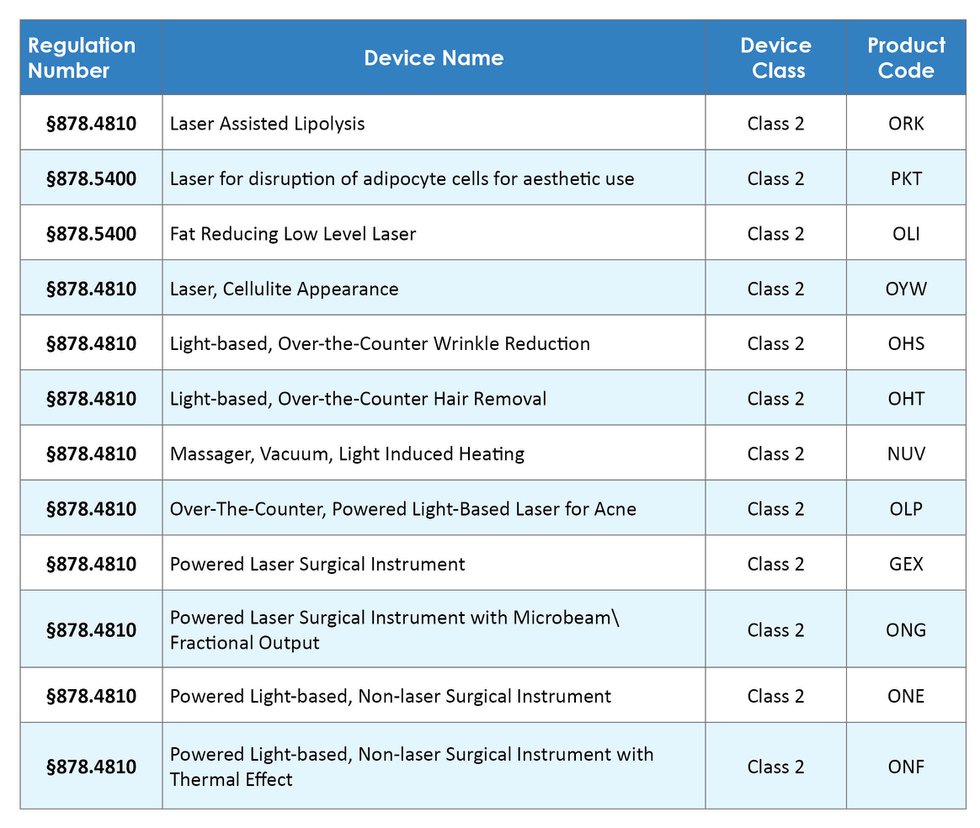
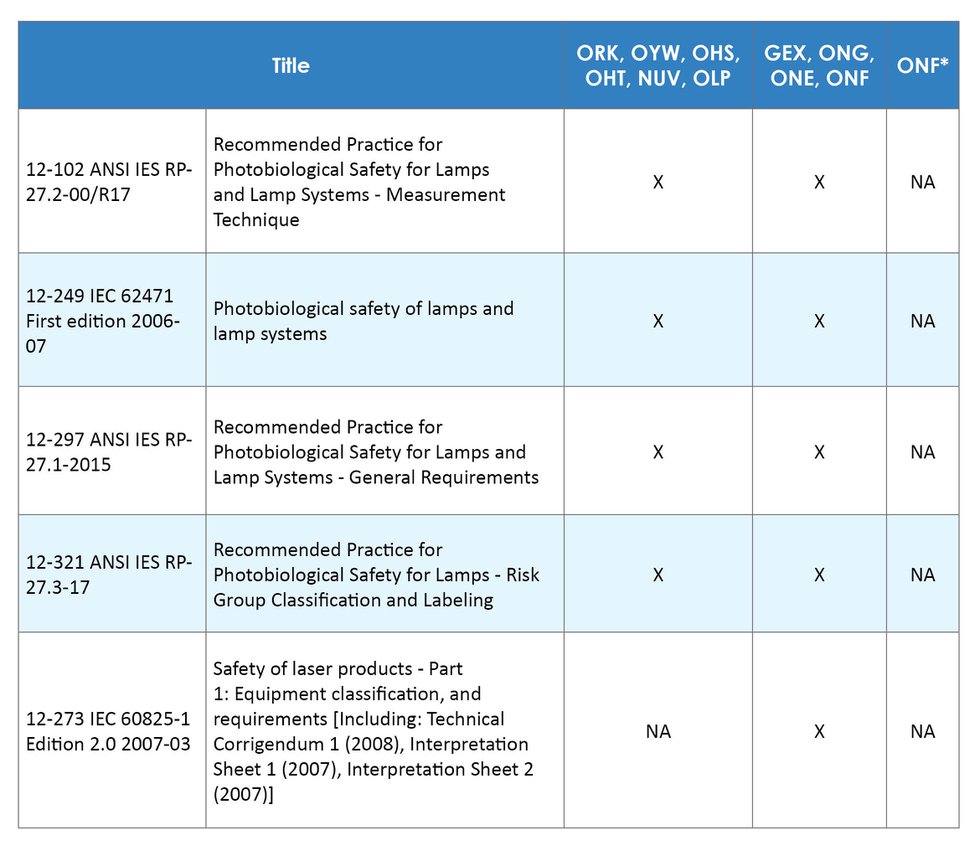
On January 20, 2023, FDA issued a final rule entitled “Radiological Health Regulations; Amendments to Records and Reports for
Laser Products - Conformance with IEC 60825-1 Ed. 3 and IEC 60601-2-22 Ed. 3.1 (Laser Notice No. 56) - Guidance for Industry and

Clarify the indications for FDA certification requirements for LED lighting products entering the US market-Industry News-Shenzhen STS test services Co., Ltd.
Are There "FDA Registered" or "FDA Certified" Medical Devices? How Do I Know What Is FDA Approved? | FDA