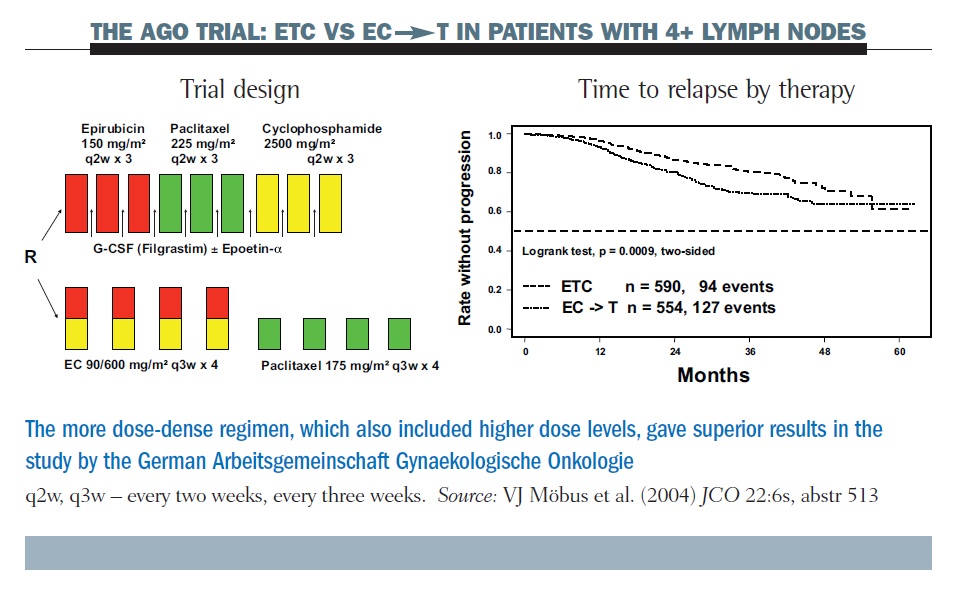
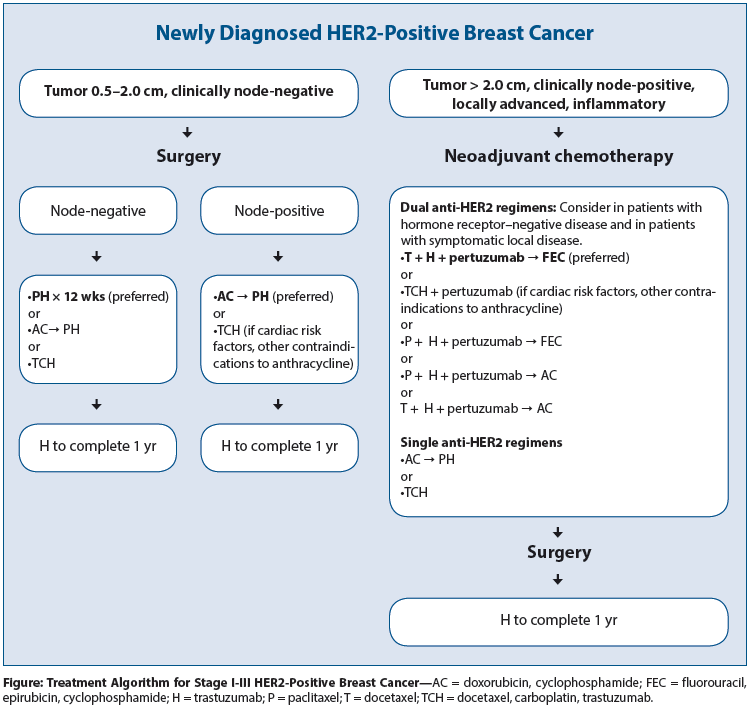
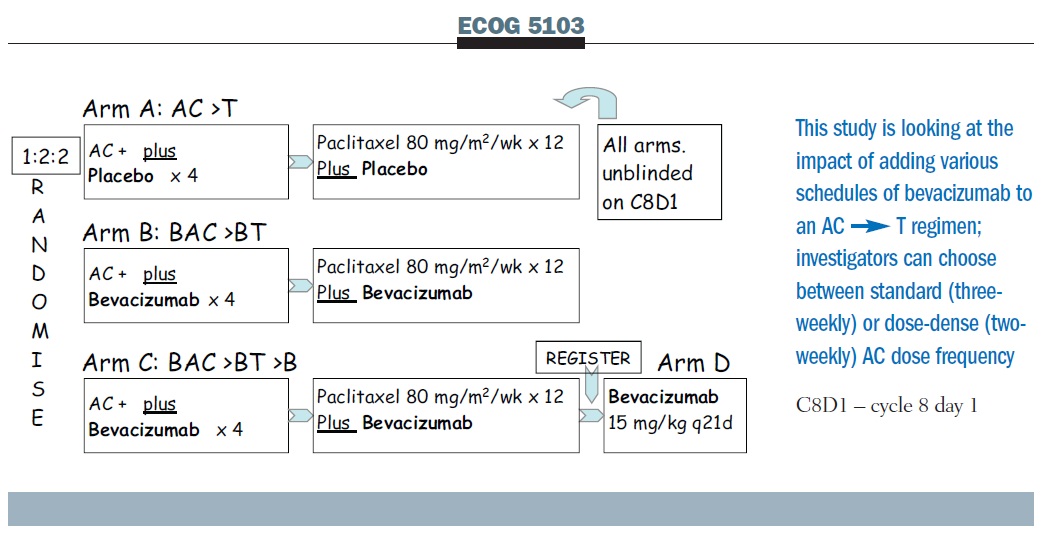
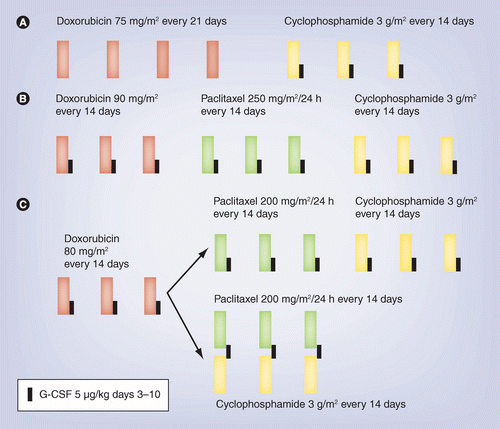
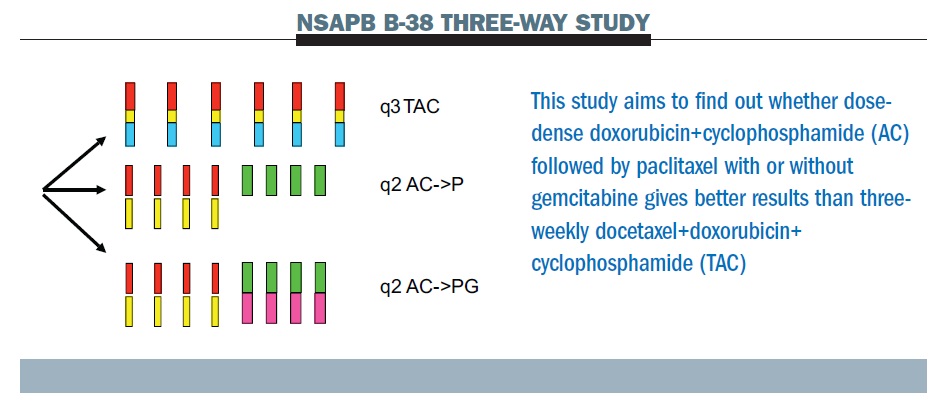
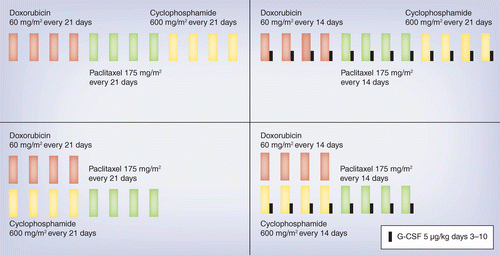
PDF) Dose Dense Doxorubicin and Cyclophosphamide Followed by Paclitaxel (Dose Dense AC Followed by T) Regimen for Breast Cancer

Standardizing Chemotherapy Regimen Nomenclature: A Proposal and Evaluation of the HemOnc and National Cancer Institute Thesaurus Regimen Content | JCO Clinical Cancer Informatics

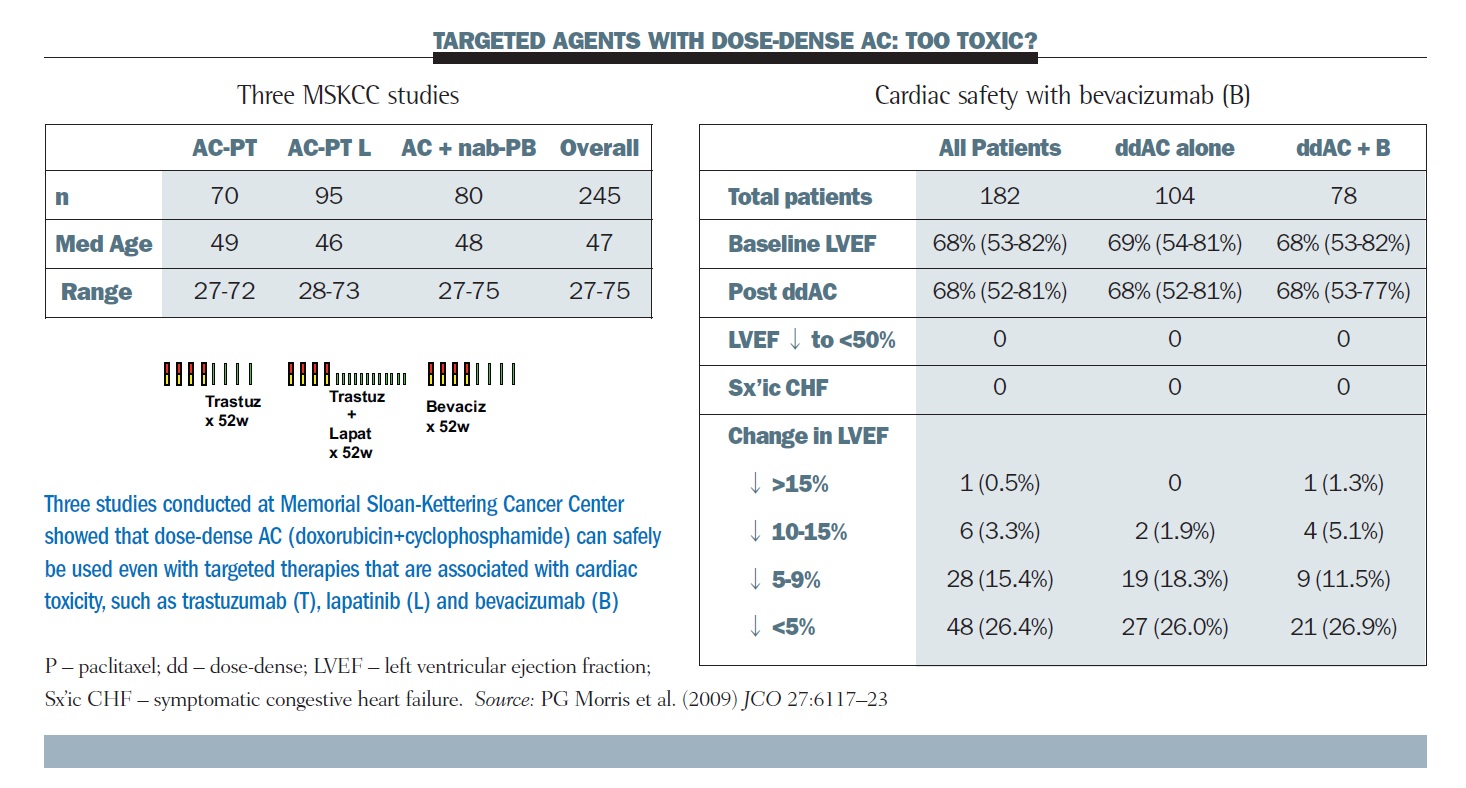
SWOG S 0800 ( NCI CDR 0000636131 ) : addition of bevacizumab to neoadjuvant nab-paclitaxel with dose-dense doxorubicin and cyclophosphamide improves pathologic complete response ( pCR ) rates in inflammatory or locally

Sequential docetaxel as adjuvant chemotherapy for early breast cancer (TACT): an open-label, phase III, randomised controlled trial - The Lancet

Anthracycline-free or short-term regimen as adjuvant chemotherapy for operable breast cancer: A phase III randomized non-inferiority trial - The Lancet Regional Health – Western Pacific

Phase 2 Study of Dose-Dense Doxorubicin and Cyclophosphamide Followed by Eribulin Mesylate With or Without Prophylactic Growth Factor for Adjuvant Treatment of Early-Stage Human Epidermal Growth Factor Receptor 2–Negative Breast Cancer -
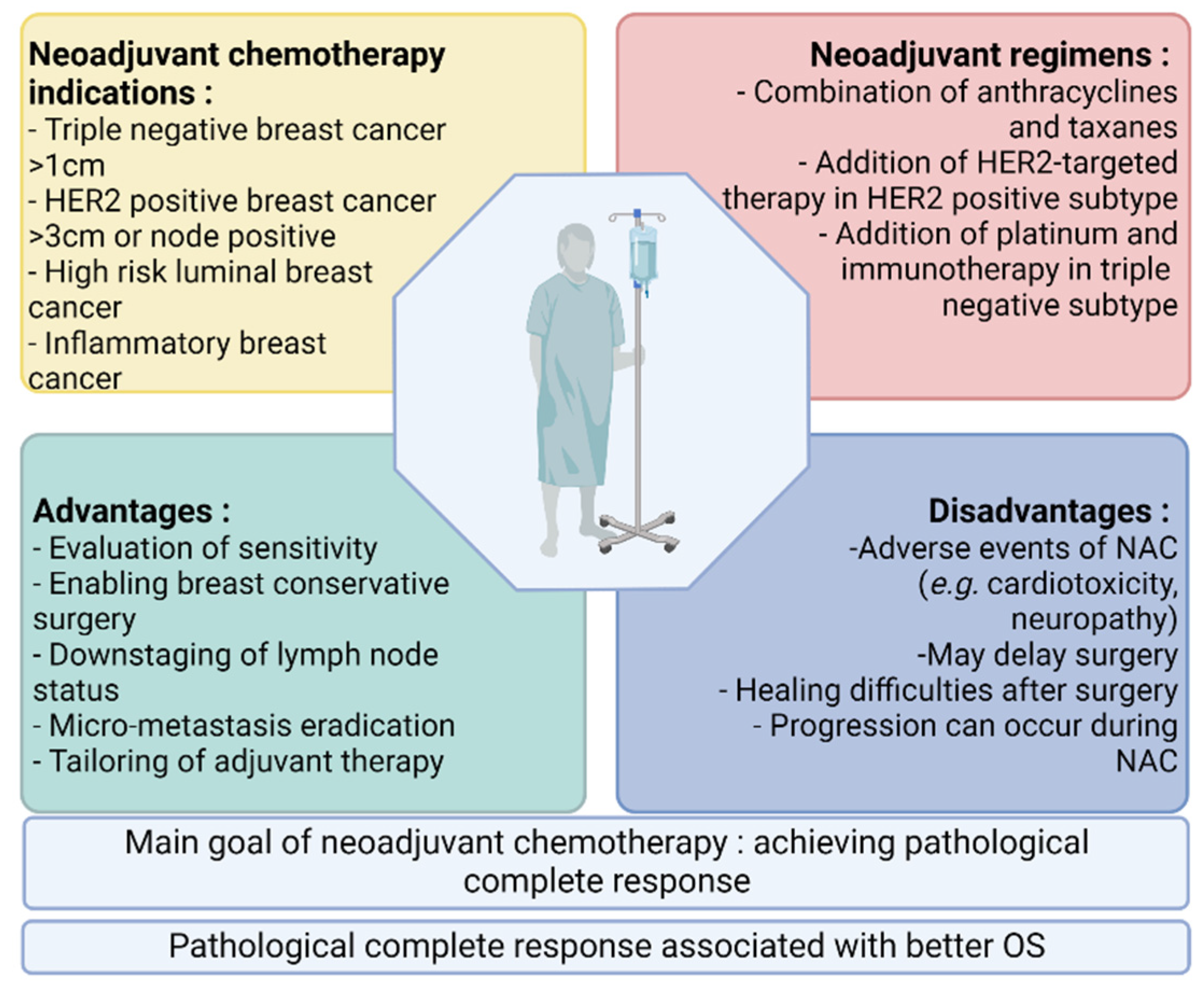
Cancers | Free Full-Text | Predictive Biomarkers of Response to Neoadjuvant Chemotherapy in Breast Cancer: Current and Future Perspectives for Precision Medicine